( 2) first reported that HSC quiescence is maintained by p21cip1/waf1 using p21−/− mice, numerous papers have found that long term-hematopoietic stem cells (LT-HSC) quiescence and activation are regulated by the genetic and epigenetic regulation of key molecules as well as by microenvironmental factors ( 3, 4). Only a small population of HSCs is required to initiate the entire hematopoietic process. Thus, understanding the molecular mechanisms regulating the self-renewal and cell fate determination of HSCs/progenitor cells is important for the development of clinical applications based on disease type and severity. All blood cell lineages are produced via functional maturation of a rare population of multipotent HSCs that can proliferate by self-renewal and differentiation. HSCs can be found in several organs, such as peripheral blood (PB), bone marrow (BM), and umbilical cord blood (UCB). Hematopoietic stem cells (HSCs) are multipotent primitive cells that can develop into all types of blood cells, including myeloid-lineage and lymphoid-lineage cells ( 1). 
Since HSCs can be differentiated into diverse progenitors including endothelial progenitors, they may be useful for constructing strategies for effective therapy. Among many adult stem cells, HSCs are a powerful tool to treat patients with diseases such as hematologic malignancies and liver disease. HSCs and HSCs-derived progenitors are promising cell sources in regenerative medicine and their contributions can be properly applied to treat pathophysiologic conditions. In the present review, we briefly summarized HSCs and their application in pathophysiologic conditions, including non-hematopoietic tissue regeneration as well as blood disorders. So far, many investigators are exploring HSCs and modified HSCs for use in clinical and basic science. Among many stem cells, the feasibility of using adult-derived hematopoietic stem cells in therapeutic approaches is very diverse, and is unarguably regarded as an important cell source in stem cell biology. A multidrug screening is identifying compounds that can regulate this process and may serve as potential agents to treat osteoporosis.Hematopoietic stem cells (HSCs) are regarded as one of essential cell sources for treating regenerative diseases. He has used age- and radiation-related bone marrow adiposity in vivo and in vitro to determine if senescence plays a role in MSC depletion due to fate conversion to adipocytes. Chandra utilizes lineage-tracing methods to accurately characterize the mesenchymal fate and identify the pathways that regulate cell fate. 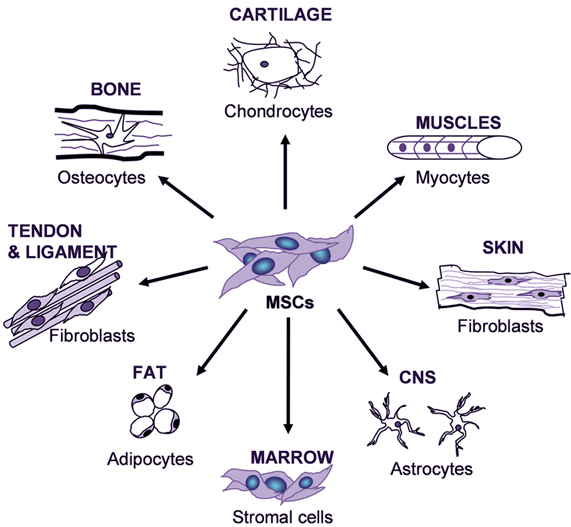
His research aims to define the cues that regulate mesenchymal cell fate and assign them to an osteogenic or adipogenic fate. Chandra focuses on therapeutic options to regulate bone marrow adiposity. This is one major reason bones fail to regenerate in osteoporotic conditions.ĭr. Intriguingly, the depletion in functional MSCs often correlates with the increase in adiposity. Bone marrow has a limited MSC pool that depletes with age and after disease. Mesenchymal stem cells (MSCs) are multipotent stem cells that can differentiate into a variety of cell types, including bone cells (osteoblasts), cartilage cells (chondrocytes), muscle cells (myocytes) and fat cells that give rise to marrow adipose tissue (adipocytes). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
